A Mines alumnus-professor pushes the envelope on laser technology.

Jeff Squier has supervised Erica Block’s research into SSTF focus for lasers, which has great potential for improving the precision and safety of laser surgery, particularly for eyes.
How can light be manipulated to slice a clean incision in the fragile lens of a human eye? Or to create three-dimensional videos of processes deep inside the brain? Or to carve a tiny laboratory on a microscopic chip?
These are not the questions Jeff Squier ’84, MS ’86 was pondering in 7th grade, when he wrote the words ‘optical physicist’ on a questionnaire asking what he wanted to be when he grew up.
“Really, I just liked the idea of playing with light,” he recalls.
Thirty-nine years later, that boyhood fascination has led the Mines physics professor and researcher to not only ask such questions, but also answer them by designing and building ultra-fast lasers that can cut, image and micro-machine in ways never before possible. His research team’s creations have been used by everyone from neuroscientists wanting to peer inside the dense neuronal tangles of a fly’s brain, to geneticists wanting to better understand, at the molecular level, how a corn plant grows. Through collaborations with private companies and other research institutions, they’re also working to develop more affordable and accurate laser-based microscopes and safer laser-based surgical techniques.
“He isn’t just building a bigger laser. He has a deep respect for the way light interacts with material,” says neuroscientist and University of California, San Diego, professor David Kleinfeld, who has used Squier’s lasers to chart blood flow and neuronal activity in the brains of rodents. “He is very clever in building lasers with interesting intrinsic properties. They could have a big impact.”
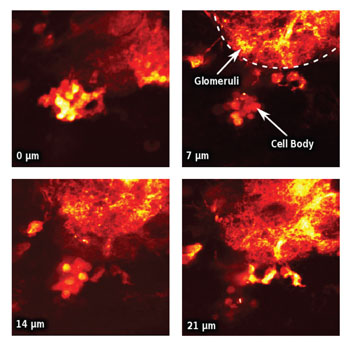
PENETRATING IMAGES: Taken simultaneously using a single detector, these four images of the interior of a fly antenna illustrate cellular activity on four parallel planes 7 micrometers apart.
A better view
The first lasers were invented in the late 1950s and, as Squier puts it, were initially ‘a solution looking for a problem.’ They moved from laboratory obscurity to pop culture fame in 1964, when the villain in the James Bond thriller ‘Goldfinger’ used a laser to gruesomely fillet his adversaries. By the ’70s, an array of industries were finding a more practical use for them.
“They captivated everyone’s attention,” Squier says. “Now they’re in everything from scanners in the supermarket, to your Blu-ray player at home, to the operating room.”
But, as he points out, not all lasers are the same.
While conventional lasers use a continuous beam of light, new ‘femtosecond’ lasers developed in the early ’80s chopped that light into infinitesimally short, ultra-intense bursts lasting just a few one-millionths of one-billionth of a second.
The possible applications of such a precise and powerful pulse of light fascinated Squier. So after earning a master’s degree in physics from Mines in 1986, he sought out one of the fathers of femtosecond lasers, G’rard Mourou at Rochester University, and went there to get his PhD in optics under his guidance.
Soon he was replacing the light bulbs in microscopes with femtosecond lasers, with remarkable results.
“It acts like the ultimate strobe camera,” he explains. “If I can make a very short burst of light, I can freeze motion and understand really fast events (like a material melting, or a chemical reaction occurring inside a cell). It can aid our understanding of a whole array of processes that have been too fast for us to follow before.”
Squier notes that laser imaging was long fraught with a perplexing shortfall. Aimed at thin-walled living cells, which don’t absorb or scatter much light, they produced a low-resolution image with little contrast. They also spread their beam broadly, creating a bleaching effect far beyond the intended focal plane. And they tended to damage the sample.
“With a normal laser everything lights up. With a femtosecond laser, only the exact focal plane lights up. This is huge,” Squier says. Because it uses an infrared beam, rather than an ultraviolet one, a femtosecond laser is also less damaging to the sample. And it can penetrate deeper, allowing for exploration of cells well below the surface.
In 1992, Squier was co-inventor of the first ever video-rate multi-photon imaging system, a laser-based device that allowed biologists to take detailed sequential photos of cell activity and string them together in a frame-by-frame animation. (The technology is widely used today). He’s been toiling in the lab ever since, working to make faster, real-time, three-dimensional, high-resolution imaging possible.
In 2012, his team took a major technological step forward when they developed a system that allows for simultaneous imaging at multiple locations. Rather than take one picture at one location of a rat brain or plant stalk and then move along to another, it can take as many as six at once from six different planes, and then splice them together to create a vivid 3D video.
Thus far, it’s not commercially available. Work will start this summer on a new system at the University of California, San Diego, where it will be used to better understand the inner workings of a fruit fly’s brain. “They are really trying to understand the neural network,” says Squier. “How does a smell get converted into an electrical signal inside the brain and what path does it follow?”

ILLUSTRATING THE POINT: The two photos depict the depth of field, or length, of a standard femtosecond focus (left) and an SSTF focus (right) projected into a fluid that fluoresces as a result of a phenomena called two-photon excitation fluorescence. The benefit of SSTF is clearly visible, as the depth of focus (length) of the fluorescence is strictly confined compared to the standard focus.
A cleaner cut
Squier has also been instrumental in shaping the femtosecond laser into an ultra-precise scalpel. He holds one of the original patents for femtosecond Lasik eye surgery.
And in May of this year, he, PhD student Erica Block, and colleague Charles Durfee, an associate professor in the Department of Physics, published a groundbreaking paper demonstrating that a technique called simultaneous spatio-temporal focusing (SSTF) could vastly improve the precision with which living biological tissue can be cut.
The complex light trick involves breaking the beam into its many colors and then lassoing them back together in a micro-instant at the focal plane.
“We focus the laser beam not just spatially but also in time,” says Squier. “It enables us to generate tremendous intensities and really precisely deliver the light in a controlled fashion.”
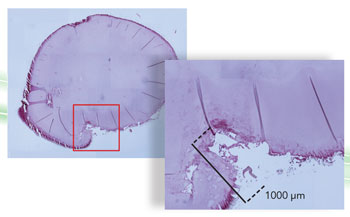
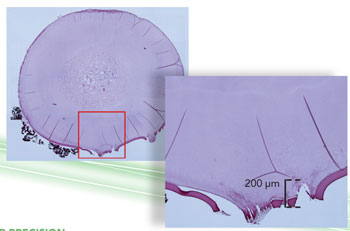
LASER PRECISION: In an experiment conducted in Squier’s lab, minute laser incisions in the lenses of pig eyes were later analyzed. The areas inside the red boxes are correspondingly magnified in the adjacent, overlapping image. The pair on the top�show the results of standard focus; the images on the bottom�show SSTF focus. In addition to achieving greater precision, SSTF focus left the thin protective membrane covering the lens mostly in place, an outcome likely to expedite healing in a human patient.
For one recent experiment, he and his colleagues made incisions in the lens of a pig’s eye, using both conventional laser technology and SSTF. The difference was stark, with the SSTF laser cutting a clean line while the conventional laser was more jagged and bubbled, a situation that in a human patient could increase risk of complications.
Thus far, eye surgery is the most obvious application. But Kleinfeld, who is collaborating with Squier to explore surgical applications, believes surgeons who operate in extremely delicate areas, like ear, nose and throat specialists, could also benefit from the technology someday.
 “It’s a way to do the cutting in a different and more efficient way,” he says.
“It’s a way to do the cutting in a different and more efficient way,” he says.
Training the next generation
Futuristic imaging and surgeries aside, Squier says he already sees immediate uses for both the cutting and imaging laser tools. The cutting tool could be used now to prototype three-dimensional glass and plastic microfluidic devices, or ‘labs-on-a-chip,’ for conducting scientific research or medical lab tests. And already, by building custom-designed laser-based microscopes for use by researchers at Mines and other institutions, his work is saving them money.
“A commercial multi-photon microscope costs about a half-million dollars right now without a laser. My students can build one with a laser for $35,000 that outperforms the commercial ones,” he says. “Plus, instead of making the experiment fit the microscope, we build the microscope to fit the experiment.”
Meanwhile, he’s giving his students an education they can get nowhere else.
“There are only a handful of groups in the world that develop the lasers and the technology around the lasers, and do experiments with them,” he says, noting that institutions elsewhere often buy the microscopes with which they conduct research. “We do everything and our students have to do everything. I think it gives them a unique skill set.”




Such As The Drop It!, this application involves the dinner and can also be free -tracking
capabilities. The iPad 2 can not be upgraded for the reason that fashion. The iPad 2 had a fantastic screen of just
one,024 x 768.
This is the very best way to be capable of give helpful advice about any item.
Getting your files saved on a independent computer like this
can arrive in useful in an unexpected emergency.