 Over the last 25 years, a series of incremental improvements to photovoltaic cells have raised efficiency levels from around 15 percent in the early ’80s to 20 percent today. However, after recent research at Mines helped confirm the effectiveness of quantum dots, scientists believe this new technology could elevate efficiency to 40 percent over the next 10 years.
Over the last 25 years, a series of incremental improvements to photovoltaic cells have raised efficiency levels from around 15 percent in the early ’80s to 20 percent today. However, after recent research at Mines helped confirm the effectiveness of quantum dots, scientists believe this new technology could elevate efficiency to 40 percent over the next 10 years.
Take a look at a solar panel on a sunny Colorado day and, if you’re like most people, you won’t see much more than a blinding glare. Mark Lusk sees wasted opportunity.
“I see that glare and feel how hot the panels on my roof get and say, ‘What a waste! We’re losing energy!'” says Lusk, a Mines physics professor and solar energy researcher, who admits to checking out his panels and their energy output more than most. On a clear day, he explains, only a fraction of the photons hitting the photovoltaic cells on his roof are converted into electricity, the rest bounce off as light or are lost as heat. On a cloudy day, or as dusk approaches, the long-wavelength, low-energy particles of light are scarcely enough to produce any juice at all. On average, just 20 percent of the sun’s rays actually get converted to energy in a contemporary solar cell.
“In terms of efficiency, there is a lot of room for improvement up there,” he says.
Fueled by a six-year, $12 million grant from the National Science Foundation, Lusk and his colleagues at the Renewable Energy Materials Research Science and Engineering Center (REMRSEC) have spent the last four years working to improve that efficiency via a complex merging of nanotechnology, quantum physics and computational wizardry known as ‘exciton engineering.’
The nascent and controversial field hinges on the manipulation of ‘excitons,’ the combination of an excited electron and the hole from which it is dislodged by an incoming photon. In conventional photovoltaic cells, the exchange is generally one-for-one; upon impact, a photon creates an exciton, which sends a highly energized electron racing into an electrical circuit.

Mark Lusk, a theoretical physicist and professor at Mines, estimates that solar panels could become twice as efficient over the next decade.
�However, by using nano-sized light-absorbing particles called ‘quantum dots,’ researchers believe they can create a micro-environment where excitons, which, for much of the day, absorb considerably more energy than is needed to get just one electron into an electrical circuit, go on to share excess energy, dislodging other electrons to create more excitons. Called multiple exciton generation (MEG), scientists now believe this approach could more than double the amount of electrical energy converted from strong sunlight on cloudless days.
Along with figuring out how to make more electricity from strong sunlight, Lusk is also exploring a parallel technology that could make better use of weak sunlight on cloudy days, when lower-energy photons produce excitons that lack the zip needed to create the required voltage. Using an outside-the-box molecular design called energy pooling, he believes they can coax several weaker excitons to combine their energy into a smaller number of higher-energy excitons, creating useful current.
“People often think of quantum mechanics as weird science,” says Lusk, seated in front of a huge monitor showing a cluster of atoms and electron clouds. “But we are exploiting just that spooky physics to engineer materials with vastly improved energy conversion efficiencies.”
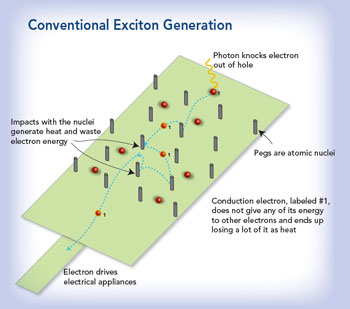
Photovoltaic cells have been around since the 1930s and still operate on basically the same principal: Particles of light, a.k.a. photons, hit the cell and nudge electrons to move to a higher energy state, “like if you had a stack of oranges and pulled one out and put it on the top, leaving a hole,” explains Lusk. The electron and its hole are together called an exciton, and they need to be sent in opposite directions through an electrical circuit. That’s the current that powers your kitchen stove or flat screen.
For years, most PV cells have been made of silicon, and improvements in efficiency have been incremental, 0.05 percent here, another 0.1 percent there. But a decade ago, scientists began to wonder if much larger strides could be made via what Lusk calls a ‘game-changing paradigm shift.’
“They said, Maybe we can take a high-energy exciton from that powerful particle of light that puts the orange way up high on the stack and somehow use some of its energy to grab another orange [electron] and pull it toward the top too, we would end up with two holes and two excited electrons out of one piece of light.” The extra energy from that powerful particle of light that would have just been wasted as heat is now turned into electrical current.
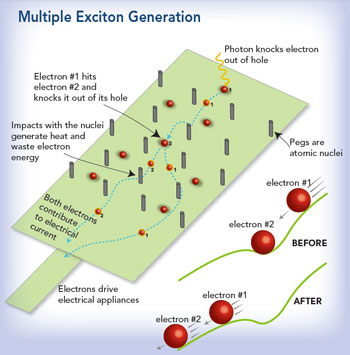
The key, researchers proposed, was to make the cell material out of infinitesimally small nano-crystals, which prompt the squeezed electron/hole pairs to behave differently than they do in bulk material, the quirky quantum confinement that Lusk loves to study.
As far back as 2000, researcher Arthur Nozik of the National Renewable Energy Laboratory (NREL) in Golden, Colo., predicted that MEG using quantum dots could boost efficiency of solar cells to as much as 65 percent, but the idea was slow to catch on.
“Most people thought it was an interesting idea, but nobody really took it seriously,” says Matt Beard, a senior scientist who arrived at NREL in 2003 and has been researching MEG ever since.
In 2004, scientists at Los Alamos National Laboratory in New Mexico became the first to actually observe MEG in action in quantum-confined materials. Soon after, some scientists were reporting that as many as seven exciton pairs could be created with one photon. But other research questioned whether nano-sizing the material made any difference.
Enter Lusk, his colleagues Alberto Franceschetti and Zhibin Lin, and a suite of high-powered computational tools, and it appears that the controversy has been settled at last. By performing a large number of ‘computational experiments,’ they were able to see exactly how electrons, holes and photons interact in quantum dots of various sizes, providing an explanation for how MEG works and why it gets better as the dots get smaller. The bottom line is that the dot size determines what excitons are most easily split up, and small dots split up their favorite excitons the best.
“It’s really unusual behavior that just comes from how we package the same old materials. The key thing is to create a thin film that, if you look closely, is full of very tiny particles,” says Lusk. Since the findings were published in a landmark paper in April 2011 in the journal ACS Nano, it’s energized the whole field of MEG research.
“We can sit here as experimentalists and measure a bunch of materials, but the problem is, first you have to make the material, and then you have to make the measurement. That takes time,” says Beard. “It is very useful when a theorist can first say, go look at X shape or Y composition. What Mark and his colleagues did at Mines was show that there is, in fact, an effect [in using quantum dots]. He helped push the theory forward.”
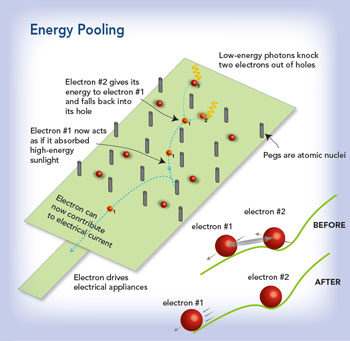
Making one where there are two
Since then, experimentalists at Mines, NREL and elsewhere have been applying the quantum dot idea to various materials, including silicon, with promising results. In December 2011, Beard published a paper in Science showing that a solar cell made of lead selenide quantum dots produced two to three electron-hole pairs per incoming photon. “It works, but we are still not there yet,” says Beard, noting that there are many steps between producing multiple excitons inside a cell and making more power.
Meanwhile, Lusk is back at the computer, working on a theoretical model of a new spider-like molecule designed to absorb lower-energy photons on its legs and launch the resulting excitons into its center, where they would pool to create a single higher-energy exciton, just the opposite of MEG. “The idea is to make a solar cell that you could put in places where there isn’t a lot of sun, or where the atmosphere is really thick,” he says.
He is also looking at ways to ‘extend the dance’ between the hole and the electron inside a quantum dot, allowing them to hop through the material together before finally splitting them apart to make current. This would make the solar cells even better because the excitons could then be pried apart in a separate piece of material that does that very efficiently. To do this, he’s taking a cue from leaves, where excitons are able to hop inexplicably long distances en route to specialized centers for making sugar.
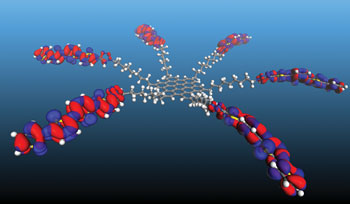
“In most materials, the dance fizzles out fast, but plant leaves somehow keep it alive,” Lusk explains. “They really have this whole quantum transport thing figured out, and I want our solar cells to do that, too.”
Elsewhere in the REMRSEC labs, scientists are taking such basic science and moving it forward, working out ways to incorporate materials made of those tiny dots into thin sheets of solar cells, which could someday be easily and cheaply applied to American homes and office buildings.
“If you look at the diversity of skills we have in this center, it’s incredible. We have physicists, chemists, materials scientists, chemical and mechanical engineers, and applied mathematicians all coming together to work on this,” says Craig Taylor, REMRSEC’s director. “We are an absolute leader in this area.”
So just how efficient could solar panels get over the next decade if this all works out?
“Forty percent is probably a really good number to shoot for now,” Lusk says. “If we could double the efficiency of solar panels, that would be like doubling the number of solar panels on the planet. Then the panels on my roof could power my house and charge an electric car. It would be huge.”
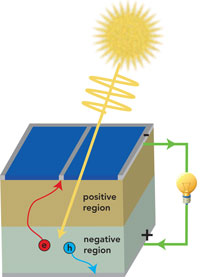 How Conventional Photovoltaic Cells Work
How Conventional Photovoltaic Cells Work
Almost all photovoltaic cells used today rely on the remarkable properties of silicon to generate electricity. When a photon from sunlight is absorbed by a silicon atom, it causes one of its electrons to move up into a higher energy level, creating a ‘hole’ on the atom where it had been residing. Electrons on neighboring atoms are able to shift over to fill this hole, leaving a new hole on the atom they just left behind. In this way, both the electron and hole are able to wander freely through the material. While the pair, together referred to as an exciton, remain linked briefly, the energetic jiggling of neighboring atoms ultimately separates them.
A silicon crystal with any number of highly energized electrons and corresponding holes jumbled together generates no electricity unless the two can be sorted. Herein lies the magic of the photovoltaic cell.
In a procedure called doping, trace amounts of particular impurities are added to silicon to create an electrostatic charge: Doping with boron causes silicon to take on a negative charge; doping with phosphorous makes it positive. If a slice of crystalline silicon is carefully doped to make the top layer positive and the bottom layer negative, an electric field forms where they meet.
This electric field performs the critical sorting function needed to generate electricity. Electrons encountering the field are swept off into the negatively charged region, while holes are swept into the positively charged region. Metallic leads then conduct electrons on the negative side into a circuit, where the current can be put to work powering electrical devices. Ultimately, the circuit returns electrons back to the positively charged side of the cell, where, having returned to a lower energy level, they recombine with the holes. �Nick Sutcliffe
Diagrams courtesy of Mark Lusk




